සැකිල්ල:Infobox drug/testcases-warning
Appearance
| This is the template test cases page for the sandbox of සැකිල්ල:Infobox drug. Purge this page to update the examples. If there are many examples of a complicated template, later ones may break due to limits in MediaWiki, see the HTML comment "NewPP limit report" in the rendered page. You can also use Special:ExpandTemplates to examine the results of template uses. You can test how this page looks in the different skins with these links: |
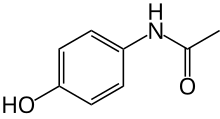
Paracetamol
[සංස්කරණය]| {{Infobox drug}} | {{Infobox drug/sandbox}} | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
References
- ^ International Drug Names
- ^ "Acetaminophen Use During Pregnancy". Drugs.com. 14 June 2019. 9 March 2020 දින පැවති මුල් පිටපත වෙතින් සංරක්ෂිත පිටපත. සම්ප්රවේශය 25 February 2020.
- ^ "Regulatory Decision Summary – Acetaminophen Injection". Health Canada. 23 October 2014. 7 June 2022 දින පැවති මුල් පිටපත වෙතින් සංරක්ෂිත පිටපත. සම්ප්රවේශය 7 June 2022.
- ^ Working Group of the Australian and New Zealand College of Anaesthetists and Faculty of Pain Medicine (2015). Schug SA, Palmer GM, Scott DA, Halliwell R, Trinca J (eds.). Acute Pain Management: Scientific Evidence (4th ed.). Melbourne: Australian and New Zealand College of Anaesthetists (ANZCA), Faculty of Pain Medicine (FPM). ISBN 978-0-9873236-7-5. 31 July 2019 දින මුල් පිටපත (PDF) වෙතින් සංරක්ෂණය කරන ලදී. සම්ප්රවේශය 28 October 2019.
- ^ a b c Forrest JA, Clements JA, Prescott LF (1982). "Clinical pharmacokinetics of paracetamol". Clin Pharmacokinet. 7 (2): 93–107. doi:10.2165/00003088-198207020-00001. PMID 7039926. S2CID 20946160.
- ^ "Codapane Forte Paracetamol and codeine phosphate product information" (PDF). TGA eBusiness Services. Alphapharm Pty Limited. 29 April 2013. 6 February 2016 දින පැවති මුල් පිටපත වෙතින් සංරක්ෂිත පිටපත. සම්ප්රවේශය 10 May 2014.
- ^ "Acetaminophen Pathway (therapeutic doses), Pharmacokinetics". 4 March 2016 දින මුල් පිටපත වෙතින් සංරක්ෂණය කරන ලදී. සම්ප්රවේශය 13 January 2016.
- ^ a b Pickering G, Macian N, Libert F, Cardot JM, Coissard S, Perovitch P, Maury M, Dubray C (September 2014). "Buccal acetaminophen provides fast analgesia: two randomized clinical trials in healthy volunteers". Drug Design, Development and Therapy. 8: 1621–1627. doi:10.2147/DDDT.S63476. PMC 4189711. PMID 25302017.
In postoperative conditions for acute pain of mild to moderate intensity, the quickest reported time to onset of analgesia with APAP is 8 minutes9 for the iv route and 37 minutes6 for the oral route.
- ^ Karthikeyan M, Glen RC, Bender A (2005). "General Melting Point Prediction Based on a Diverse Compound Data Set and Artificial Neural Networks". Journal of Chemical Information and Modeling. 45 (3): 581–590. doi:10.1021/ci0500132. PMID 15921448.
- ^ "melting point data for paracetamol". Lxsrv7.oru.edu. 30 June 2012 දින මුල් පිටපත වෙතින් සංරක්ෂණය කරන ලදී. සම්ප්රවේශය 19 March 2011.
- ^ a b c d e Granberg RA, Rasmuson AC (1999). "Solubility of paracetamol in pure solvents". Journal of Chemical & Engineering Data. 44 (6): 1391–95. doi:10.1021/je990124v.
Brincidofovir
[සංස්කරණය]| {{Infobox drug}} | {{Infobox drug/sandbox}} | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
References
- ^ "FDA approves drug to treat smallpox". U.S. Food and Drug Administration (FDA). 4 June 2021. 8 June 2021 දින පැවති මුල් පිටපත වෙතින් සංරක්ෂිත පිටපත. සම්ප්රවේශය 7 June 2021.
 මෙම ලිපිය තුළ මෙම ප්රභවයේ පෙළ ඇතුළත් වන අතර එය පොදු වසම තුළ පවතියි.
මෙම ලිපිය තුළ මෙම ප්රභවයේ පෙළ ඇතුළත් වන අතර එය පොදු වසම තුළ පවතියි.
Albendazole
[සංස්කරණය]| {{Infobox drug}} | {{Infobox drug/sandbox}} | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
References
- ^ Plumb DC (2011). "Albendazole". Plumb's Veterinary Drug Handbook (7th ed.). Stockholm, Wisconsin; Ames, Iowa: Wiley. pp. 19–21. ISBN 978-0-470-95964-0.
- ^ a b c d "Albenza, (albendazole) dosing, indications, interactions, adverse effects, and more". Medscape Reference. WebMD. March 1, 2014 දින පැවති මුල් පිටපත වෙතින් සංරක්ෂිත පිටපත. සම්ප්රවේශය February 25, 2014.