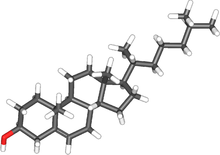
කොලෙස්ටරෝල්
| |
| |
| Names | |
|---|---|
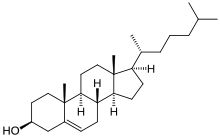
| IUPAC name
(3β)-cholest-5-en-3-ol
| |
| වෙනත් නාම
(10R,13R)-10,13-dimethyl-17-(6-methylheptan-2-yl)-2,3,4,7,8,9,11,12,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-3-ol
| |
| Identifiers | |
| CAS number | 57-88-5 |
| PubChem | 5997 |
| ChemSpider | 5775 |
| SMILES |
|
| Properties | |
| Molecular formula | C27H46O |
| අණුක ස්කන්ධය | 386.654[1] |
| Appearance | white crystalline powder [2] |
| Melting point |
148–150 °C [2] |
| Boiling point |
360 °C (decomposes) |
| Solubility in water | 0.095 mg/L (30 °C) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |

කොලෙස්ටරෝල්(Cholesterol) යනු ලිපිඩමය ඇලෙනසුලු ඇල්කොහොල් විශේෂයකි.මෙය සතුන් ගේ පටකය හා රුධිර ප්ලාස්මාවේ අඩංගුය.එය ක්ෂීරපායින්ගේ සෛල තුල අඩංගු වැදගත් අංගයකි සෛල ප්ලාස්මයේ පාරගම්යතාවය හා තරලතාවය පවත්වාගැනිමට උපකාරවෙයි.සත්ව දේහයේ ඇති ප්රධාන ස්ටෙරෝල් සංයෝගය මෙයයි,එහෙත් වෙනත් සෛලමය දේහයන් තුලද කුඩා ප්රමාණයක් කොලෙස්ටරෝල් අංගුය, උදාහරණ ලෙස ශාක හා දිලීර වැනි රාජධානීන්ට අයත් ජීවීන් ගේ සෛල දැක්විය හැක.මෙය බැක්ටීරියා තුල කොලෙස්ටරෝල් අඩංගු නැත.[3] කොලෙස්ටරෝල් ස්ටෙරොයිඩ් කාණ්ඩයට අයත් රසායනික සංයෝගයකි.
කොලෙස්ටරෝල් ශරීරයට අත්යවශ්ය වුවද අධික කොලෙස්ටරෝල් මට්ටම ශරීරයට අහිතකරය.ශරීරයට අවශ්ය කොළෙස්ටරෝල් ප්රමාණය ආහාර මගින් ශරීරයට අවශෝෂණය කරගනී.
කොළෙස්ටරෝල් යන වචනය ග්රීක භාෂාවේ chole හා stereos යන වචන ඇසුරින් සෑදුන වචනයකි,1815 දී රසායන විද්යඥයෙකු වූ ඉයුජන් චෙවෙයුල් විසින් මෙය කොළෙස්ටරෝලි ලෙස නම්කළේය.[4]
කායික විද්යාව[සංස්කරණය]
කෘත්ය[සංස්කරණය]
මෙම කොටස හිස්ය. එය පුළුල් කිරීමෙන් ඔබ හට උපකාර කළ හැක. |
සංයෝගය හා අවශෝෂණය[සංස්කරණය]
මෙම කොටස හිස්ය. එය පුළුල් කිරීමෙන් ඔබ හට උපකාර කළ හැක. |
ප්ලාස්මා ප්රවාහය[සංස්කරණය]
මෙම කොටස හිස්ය. එය පුළුල් කිරීමෙන් ඔබ හට උපකාර කළ හැක. |
සමස්ථිතිය[සංස්කරණය]
මෙම කොටස හිස්ය. එය පුළුල් කිරීමෙන් ඔබ හට උපකාර කළ හැක. |
පරිවෘතිය හා බහිස්ස්රාවය[සංස්කරණය]
මෙම කොටස හිස්ය. එය පුළුල් කිරීමෙන් ඔබ හට උපකාර කළ හැක. |
ආහාර ප්රභවය[සංස්කරණය]
මෙම කොටස හිස්ය. එය පුළුල් කිරීමෙන් ඔබ හට උපකාර කළ හැක. |
සායනික වැදගත්කම[සංස්කරණය]
මෙම කොටස හිස්ය. එය පුළුල් කිරීමෙන් ඔබ හට උපකාර කළ හැක. |
අමතර රූප[සංස්කරණය]
-
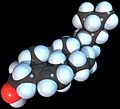
Space-filling model of the Cholesterol molecule
-
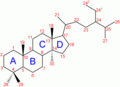
Numbering of the steroid nuclei
සටහන්[සංස්කරණය]
- ↑ සැකිල්ල:PubChem
- ↑ 2.0 2.1 "Safety (MSDS) data for cholesterol". සම්ප්රවේශය 2007-10-20.
- ↑ Pearson A, Budin M, Brocks JJ (2003). "Phylogenetic and biochemical evidence for sterol synthesis in the bacterium Gemmata obscuriglobus". Proc. Natl. Acad. Sci. U.S.A. 100 (26): 15352–7. doi:10.1073/pnas.2536559100. PMC 307571. PMID 14660793.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ↑ Olson RE (1998). "Discovery of the lipoproteins, their role in fat transport and their significance as risk factors". J. Nutr. 128 (2 Suppl): 439S–443S. PMID 9478044.
{{cite journal}}: Unknown parameter|day=ignored (help); Unknown parameter|month=ignored (help)
බාහිර යොමු[සංස්කරණය]
- Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults US National Institutes of Health Adult Treatment Panel III
- Aspects of fat digestion and metabolism – UN/WHO Report 1994
- American Heart Association – "About Cholesterol"